1 Overview
The male germ line produces only male gametes, spermatids. As in the hermaphrodite, the male germ line exhibits distal-proximal polarity with a mitotic cell population located at the most distal end of the gonad (DG) and meiotic cells extending proximally. Gametogenesis occurs in the proximal germ line and the resulting spermatids are stored in the seminal vesicle until ejaculation (MaleReproFIG 17A; Hirsh et al., 1976; Klass et al., 1976; Kimble and Hirsh, 1979).
MaleReproFIG 17A: Adult male germ line organization. Nomarski DIC picture of an adult male tail region featuring the parts of the reproductive tract, lateral view. (DG) Distal gonad; (PG) proximal gonad; (DTC) distal tip cell.
Much of the male germ line is a syncytium. Germ cells have incomplete borders and are connected to one another via a central canal, the rachis (MaleReproFIG 17B and 17C). The germ line is ensheathed only by the gonadal basal lamina (gbl) which also covers the somatic portion of the gonad.
MaleReproFIG 17B: Ultrastructure of the male germ line. TEM shows cross section of both distal and proximal arms of the germ line, transverse view. (Image source: him5-25 [Hall] 9112.) (Gbl) Gonadal basal lamina.
MaleReproFIG 17C: Ultrastructure of the male germ line. TEM shows cross section of proximal arm of the germ line lying adjacent to the intestine, transverse view. (Image source: him5-25 [Hall] 0702.) (Gbl) Gonadal basal lamina.
2 Mitosis in the Germ Line
As in the hermaphrodite, the distal end of the male germ line (MaleReproFIG 18A) contains a stem cell population and is referred to as the mitotic zone. As germ cells move away from the influence of the distal tip cell (DTC, described in Male Reproductive System - Somatic Gonad), they enter meiosis I, proceeding through prophase I to pachytene (Hirsh et al., 1976). Nuclear morphology at various stages of mitosis and meiosis I can visualized with DAPI staining (examples from the hermaphrodite germ line, highlighting stage-specific characteristics, are shown in MaleReproFIG 18B-D).
MaleReproFIG 18: Cell division in the germ line. A. Nomarski DIC image shows locations of mitosis, meiosis and gametogenesis in the germ line. B-D. DAPI -stained germ line cells. (Image source: J. Maciejowski and E.J. Hubbard). B. Mitosis occurs at the distal end of the germ line in an area called the mitotic zone. C. As germ cells move farther away from the distal tip cell (DTC), they enter an area called the transition zone where they enter meiosis I. D. Cells proceed through prophase I to pachytene. Chromosomes spread out along the periphery of the nucleus forming a "bowl of spaghetti" phenotype.
3 Spermatogenesis and Spermiogenesis
See Sperm Gallery for detailed pictures and videos
After pachytene, the precursor of spermatids, the spermatocytes, detach from the rachis and complete meiosis generating haploid spermatids (MaleReproFIG 19 and 20). This process of spermatid formation is called spermatogenesis. Spermatids are stored in the seminal vesicle until ejaculation. Spermiogenesis, activation of sessile spermatids into crawling spermatozoa, occurs after mating, in the hermaphrodite uterus (see Spermiogenesis Figure and Video). Some spermatids that remain in the male after mating, however, may mature into spermatozoa and can be seen swimming around the vas deferens and seminal vesicle (Ward and Carrel, 1979). Recent studies suggest that the male gonad contains substances that prevent premature activation of spermatids prior to sperm transfer (Stanfield and Villeneuve, 2004).
MaleReproFIG 19: Sperm development. Illustration showing the stages of spermatogenesis and spermiogenesis resulting the production of motile sperm. (Based on L'Hernault, 1997.)
|
|
MaleReproFIG 20: Male sperm production. Nomarski DIC picture showing the location of spermatocytes and spermatids within the adult male germ line, lateral view.
Developing spermatocytes contain a large number of specialized vesicles called FB-MOs (Fibrous Body-Membranous Organelle) (MaleReproFIG 21, 22 and 23). These organelles contain proteins required in the future spermatid and spermatozoon, including MSP (Major Sperm Protein; Wolf et al., 1978; Ward and Klass,1982). During development, the FB-MOs partition with the portion of the spermatocyte destined to become the future spermatid (Ward, 1986). The residual body (MaleReproFIG 22; Dividing Spermatocytes Figure) acts as a depot for proteins and organelles no longer required by the developing spermatid (L'Hernault, 1997; Arduengo et al., 1998; Kelleher et al., 2000).
MaleReproFIG 21: FB-MOs production. Panel on left shows the process of FB-MO (fibrous body membranous organelle) formation. Right panel illustrates the parts of the FB-MO. (Based on Arduengo et al., 1998.)
Maturing spermatids and spermatozoa have highly condensed nuclei (N) and tightly packed mitochondria (M) (MaleReproFIG 23; Spermatozoon TEM Figure). In spermatids, MOs (now lacking FB) locate near the cell periphery. During spermatid activation, MOs fuse with the plasma membrane releasing their contents (primarily glycoproteins) onto the cell surface. A fusion pore is generated on the cell surface by the MO collar (MaleReproFIG 24). Mutants affected in MO fusion produce sperm with defective motility suggesting that MO content enhances sperm mobility (Ward et al., 1981; Roberts et al, 1986; Achanzar and Ward, 1997). Spermatid activation also involves the formation of a foot or pseudopodium (MaleReproFIG 24; Spermatozoon SEM Figure). In contrast to sperm in other phyla, C. elegans sperm lack flagella. Pseudopodia allow spermatozoa to attach to the walls of the spermathecal or seminal vesicle lumen and to crawl (see Crawling Sperm Video). This motility is driven by dynamic polymerization of MSP (Major Sperm Protein). In addition to an intracellular cytoskeletal function, MSP contains sequences that mediate extracellular signaling (see Oocyte maturation section of Hermaphrodite Reproductive System - Germ Line; Miller et al, 2001; Italiano et al., 1996; Roberts and Stewart, 2000).
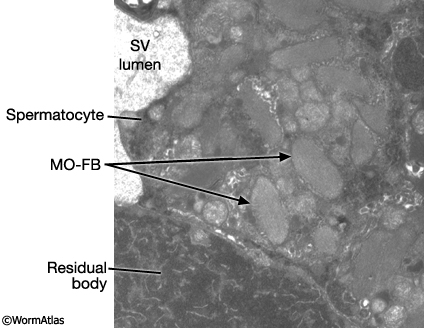
MaleReproFIG 22: Ultrastructure of FB-MO. TEM of FB-MO in developing spermatocytes, transverse view. (Image source: him5-25 [Hall] 1215.) (SV) Seminal vesicle.
MaleReproFIG 23: Ultrastructure of FB-MO in maturing sperm. TEM of FB-MO in maturing spermatids and spermatozoa showing the highly condensed nuclei and tightly packed mitochondria, transverse view. The collar region of the FB-MO is indicated by white arrows. (Image source: him5-25 [Hall] 2631.) (SV) Seminal vesicle.
|
MaleReproFIG 24: Ultrastructure of spermatozoon. TEM of spermatid activation. The MO fuses with the plasma membrane via the collar. Pseudopodia can all be seen. (Image source: him5-25 [Hall] H707.)
4 Germ-line Development
Male germ line development spans L1 to early adulthood. All germ cells are descended from either Z2 or Z3 (Kimble and Hirsh, 1979). Key events in germ line development are summarized in MaleReproFIG 25. In contrast to somatic lineage development in C. elegans, germ line cell divisions appear to be variable with respect to their timing and planes of division (Kimble and Hirsh, 1979) and hence the precise lineal relationships between these cells are not known.
MaleReproFIG 25: Male germ line development. Illustration showing the stages of germ line formation in the developing male. (Gbl) Gonadal basal lamina; (DTC) distal tip cell; (SPm) somatic gonadal primordium of the male; (DG) distal gonad; (PG) proximal gonad. (Adapted from Schedl, 1997.)
5 References
Achanzar, W.E. and Ward, S. 1997. A nematode gene required for sperm vesicle fusion. J. Cell Sci. 110: 1073-1081. Article
Arduengo, P.M., Appleberry, O.K., Chuang, P. and L'Hernault, S.W. 1998. The presenilin protein family member SPE-4 localizes to an ER/Golgi derived organelle and is required for proper cytoplasmic partitioning during Caenorhabditis elegans spermatogenesis. J. Cell Sci. 111: 3645-3654. Article
Hirsh, D., Oppenheim, D. and Klass, M. 1976. Development of the reproductive system of Caenorhabditis elegans. Dev. Biol. 49: 200-219. Abstract
Italiano, J.E. Jr., Roberts, T.M., Stewart, M. and Fontana, C.A. 1996. Reconstitution in vitro of the motile apparatus from the amoeboid sperm of Ascaris shows that filament assembly and bundling move membranes. Cell 84: 105-114. Article
Kelleher, J.F., Mandell, M.A., Moulder, G., Hill, K.L., L'Hernault, S.W., Barstead, R. and Titus, M.A. 2000. Myosin VI is required for asymmetric segregation of cellular components during C. elegans spermatogenesis. Curr. Biol. 10: 1489-1496. Article
Kimble, J. and Hirsh, D. 1979. The postembryonic cell lineages of the hermaphrodite and male gonads in Caenorhabditis elegans. Dev. Biol. 70: 396-417. Article
Klass, M., Wolf, N. and Hirsh, D. 1976. Development of the male reproductive system and sexual transformation in the nematode C. elegans. Dev. Biol. 52: 1-18. Abstract
L'Hernault, S.W. 1997. Spermatogenesis. In C. elegans II (ed. D. L. Riddle et al.). Chap. 11. pp. 417-500. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York. Article
Miller, M.A., Nguyen, V.Q., Lee, M.H., Kosinski, M., Schedl, T., Caprioli, R.M. and Greenstein, D. 2001. A sperm cytoskeletal protein that signals oocyte meiotic maturation and ovulation. Science 291: 2144-2147. Abstract
Roberts, T.M. and Stewart, M. 2000. Acting like actin. The dynamics of the nematode major sperm protein (msp) cytoskeleton indicate a push-pull mechanism for amoeboid cell motility. J. Cell Biol. 149: 7-12. Article
Roberts, T.M., Pavalko, F.M. and Ward, S. 1986. Membrane and cytoplasmic proteins are transported in the same organelle complex during nematode spermatogenesis. J. Cell Biol. 102: 1787-1796. Article
Schedl, T. 1997. Developmental Genetics of the Germ Line. In C. elegans II (ed. D. L. Riddle et al.). chap. 10. pp. 417-500. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York. Article
Stanfield, G.M. and Villeneuve, A.M. 2006. Regulation of sperm activation by SWM-1 is required for reproductive success of C. elegans males. Curr. Biol. 16: 252-63. Article
Ward, S. 1986. Asymmetric localization of gene products during the development of C. elegans spermatozoa. In Gametogenesis and the Early Embryo. (ed. J. G. Gall). pp. 55-75. 44th Symposium of the Society for Developmental Biology. Alan R. Liss, New York.
Ward, S. and Carrel, J.S. 1979. Fertilization and sperm competition in the nematode Caenorhabditis elegans. Dev. Biol. 73: 304-321. Abstract
Ward, S. and Klass, M. 1982. The location of the major protein in Caenorhabditis elegans sperm and spermatocytes. Dev. Biol. 92: 203-208. Abstract
Ward, S., Argon, Y. and Nelson, G.A. 1981. Sperm morphogenesis in wild-type and fertilization-defective mutants of Caenorhabditis elegans. J. Cell Biol. 91: 26-44. Article
Wolf, N., Hirsh, D. and McIntosh, J.R. 1978. Spermatogenesis in males of the free-living nematode, Caenorhabditis elegans. J. Ultrastruct. Res. 63: 155-69. Abstract
|