|
ALIMENTARY SYSTEM OF THE MALE - PROCTODEUM
 Click pictures for new window with figure and legend Click pictures for new window with figure and legend
1 General Information
The proctodeum of the male is essentially a modified rectum. In contrast to the hermaphrodite, the male rectum is an outflow for the genital, as well as, the alimentary tract. In addition, it houses structures required for copulation. Tissues that contribute to the proctodeum include the rectal valve and rectal glands, the cloaca, the copulatory spicules, the spicule channels and the gubernaculum (MaleProcFIG 1). The rectal valve and glands are present in alimentary tracts of larval animals and adult hermaphrodites. The cloaca, the spicules and spicule channels and the gubernaculum are generated post-embryonically only in the male from the rectal epithelial cells that form the simple rectum of larvae and adult hermaphrodites (Sulston and Horvitz, 1977; Sulston et al., 1980).
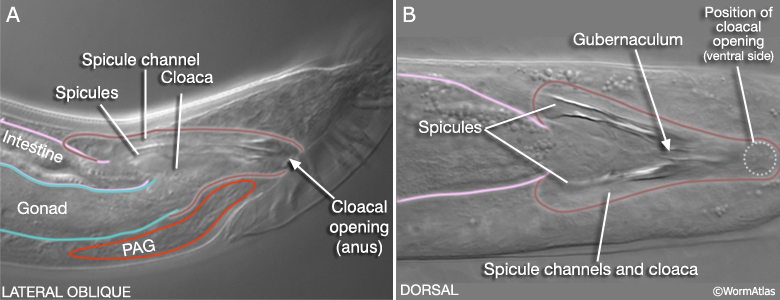
MaleProcFIG 1: The adult male proctodeum. A. Diagram showing the lateral view. (PAG) Pre-anal ganglia; (Sph) sphincter muscle. B. Slice from A shown in "end-on" view of anterior end, just posterior to the rectal glands and gonad junction.
The cloaca is an epithelial chamber that links the genital and alimentary tracts to the exterior via the anus (cloacal opening). The copulatory spicules are a left/right pair of prong-like structures housed within the cloaca that are used by the male during mating (MaleProcFIG 2; Liu and Sternberg, 1995; Garcia et al, 2001). Each consists of the dendritic processes of 2 sensory neurons surrounded by support cells (2 SPsh sheath and 4 SPso socket cells) and a thick layer of sclerotized (electron dense) cuticle. The spicules enter the sides of the cloaca anterior of the anus, via epithelial tubes called the spicule channels. The channel and cloacal walls are formed by 31-32 epithelial cells (see Cell List). Channel and cloacal lumens are lined with a continuous cuticle. During mating the spicules protrude through the cloacal opening to probe for the vulval opening or to hold the vulva open during ejaculation. The gubernaculum, a sclerotic V-shaped strip of cuticle on the cloaca's dorsal roof, deflects movement of the spicules ventrally through the cloacal opening (MaleProcFIG 2). In this chapter we focus on development and organization of the cloaca and the spicule channels. For a description of spicule development and structure see Neuronal Support Cells - The Spicules.
2 Proctodeum development
The cloaca and spicule channels are formed post-embryonically from the progeny of epithelial blast cells B, Y, F, U, K and P12 (Sulston and Horvitz, 1977; Sulston et al., 1980). The B lineage contributes 19 cells (and also produces the cells of the spicules - see Neuronal Support Cells - The Spicules), the F lineage provides 4 cells and the Y lineage 2 cells (MaleProcFIG 4). The ventral anterior portion of the cloaca forms an extensive region of contact with the gonad and this connection is formed by the daughter of K (K.a), its right partner K', 3-4 U progeny and P12.pa (hyp12) (MaleProcFIG 3, 4 and 5).
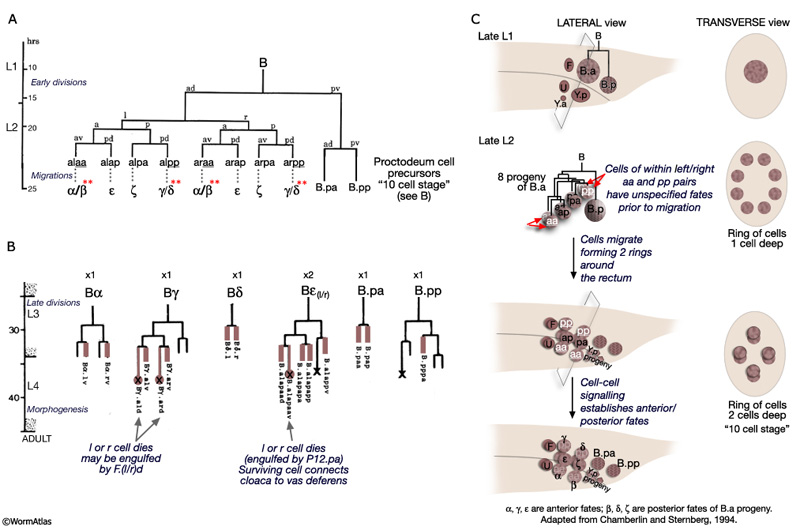
MaleProcFIG 3: Lineal origin of proctodeal cells. Diagram showing lateral view of proctodeum.
MaleProcFIG 4: F, Y, U, K', K and P12 cells contribute to the proctodeum. Letters in parentheses refer to designation used in Sulston et al., 1980. PChL/R are also part of the post-cloacal sensilla, as are other cells generated by Y. *U.ra or U.la may not divide and, instead, may fuse with the respective U.l/rp cell. (Based on Sulston and Horvitz, 1977; Sulston et al., 1980.) |
2.1 Cloaca and spicule channel cell generation
The B lineage contributes the greatest number of cells to the proctodeum. The B cell generates 47 progeny which are incorporated into either the cloaca, the spicule channels or the spicules. The B cell lineage represents one of the few cases in C. elegans where cell fate is not entirely fixed by lineal origin and is determined by environmental cues (reviewed in WormBook: Male Development - Emmons and Sternberg, 1997). This lineage is defined by three distinct phases: early division, short range migrations and late divisions (MaleProcFIG 5A&B). The early divisions begin in late L1 where the B cell divides asymmetrically to produce a large anterior daughter (B.a) and a small posterior daughter (B.p). This asymmetry is regulated by the Wnt pathway (a LIN-44 /LIN-17 pathway; Sternberg and Horvitz, 1988; Herman and Horvitz, 1994). By mid-L2 B.a and B.p produce a total of 10 cells (MaleProcFIG 5C): B.a generates 4 cell pairs and B.p 1 pair. The 4 B.a-derived pairs migrate a short distance, arranging themselves in 2 rings of 4 cells around the developing cloaca. Within the aa(l/r) and pp(l/r) left/right pairs migration of the left and right cell is variable; either can take the more anterior position near the developing cloaca. Signals from neighboring cells of the F, U (EGF LIN-3/LET-23 signaling) and Y lineages (LIN-15) promote differences between these otherwise equivalent cells causing them adopt an anterior fate (for aa's = α, for pp's=γ) or a posterior fate (for aa's = β; for pp's= δ) (distinguished by the lineage produced in later cell divisions; Chamberlin and Sternberg, 1993; 1994). Cells of the B.p lineage are patterned by Notch signaling (LIN-12) (MaleProcFIG 5C; Chamberlin and Sternberg, 1994). During L3, precursors α, δ, ε, γ, B.pp and B.pa produce the cells of the cloaca and channels. α, β, ζ and γ also generate the cells of the spicules (MaleProcFIG 5B; see Neuronal Support Cells - The Spicules).
2.2 Cloaca and spicule channel morphogenesis
The final form of the proctodeum is generated by a combination of cell division patterns, cell migration and morphogenesis. Terminal cells are generated by the L3/L4 molt (MaleProcFIG 5B). During L4, cells move progressively to their final position. Some remain near their birthplace while others migrate to new locations. For a given cell however movement and final location are invariant (MaleProcFIG 6). During late L4 there is further movement due to stretching of the entire proctodeum anteriorly as part of the process of tail morphogenesis which begins mid-L4 (ca. 40hrs) with retraction of the tail-tip and ends by the L4/adult molt (45hrs) (described in Male Epithelial System - Hypodermis). Proctodeal elongation is facilitated by interactions between dorsal spicule channel cells and dorsal body wall. Specifically, B.a(l/r)appv (Bε[l/r].pv) insert "fingers" into the dorsal spicule retractor (dsr) muscles, which are attached to the body wall hyp (MaleProcFIG 7). During tail morphogenesis the body hyp migrates anteriorward and with it drags the attached muscles and associated proctodeum. This movement also facilitates elongation of the spicules and gubernaculum (see Neuronal Support Cells - The Spicules). Ablation of the muscle blast cell M (which generates the retractor muscles) results in a compressed proctodeum containing crumpled spicules and gubernaculum. During morphogenesis there are also some programmed cell deaths. Asymmetric cell deaths occur among two left and right cell pairs of the B lineage (indicated as a circle and cross in MaleProcFIG 5B; MaleProcFIG 6): ventrally located Bεl.aav or Bεr.aav is engulfed and killed by P12.pa; dorsally located Bγ.ald or Bγ.ard is engulfed by the F.rd-F.ld syncytium (MaleProcFIG 8B). Cells move medially before dying and so surviving cells are located near the mid-line. It has been suggested that the two cells of each pair have to compete for the fate (Sulston et al., 1980).
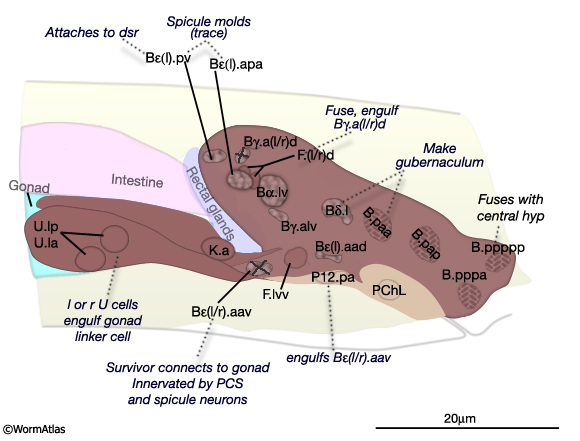
MaleProcFIG 6: Left and central nuclei of the proctodeum and associated tissues. Diagram showing lateral view of proctodeum nuclei position in late L4 stage (42 hours, 20°C). Fates and potential function of cells are indicated. Positions are largely invariant by this stage. B.a(l/r)ap=Bε(l/r); "X", left or right cell dies. (PCS) Post-cloacal sensilla. Rectal valve cells not shown. (Adapted from Sulston et al., 1980.)
MaleProcFIG 7: Proctodeum morphogenesis. Bε = B.a(l/r)ap. (dsr) Dorsal retractor muscle; (SPso) spicule socket cell. (Based on Sulston et al., 1980; Baird and Ellazar, 1999.)
During L4, B, U and K descendents of the ventral cloaca begin to establish a connection with the gonad. U progeny (U.lp and U.rp, or their fusion products), K.a and K' advance anteriorly along the outsides of the gonad, which is growing towards them (MaleProcFIG 8). As the most posterior cell of the gonad, the linker cell, passes between the U cells, one of these cells engulfs it. U and K cells attach to the gonad laterally, while either Bεl.aav or Bεr.aav meets the gonad ventrally (MaleProcFIG 10A). If U is ablated, K cells have no affinity for the gonad and the connection is not made (Sulston et al., 1980).
MaleProcFIG 8: Cloacal-gonad connection.
3 Proctodeum Structure
3.1 Proctodeum of the Late L4 Male
The arrangement of cells that make up the cloaca and spicule channels is most clearly seen during late morphogenesis, after the lineages are completed but before the final molt (MaleProcFIG 9, 10). At this time, terminal cells are filled with ribosomes and ER, as are those of surrounding tissues which are similarly undergoing the final stages of differentiation and morphogenesis (Sulston et al., 1980).
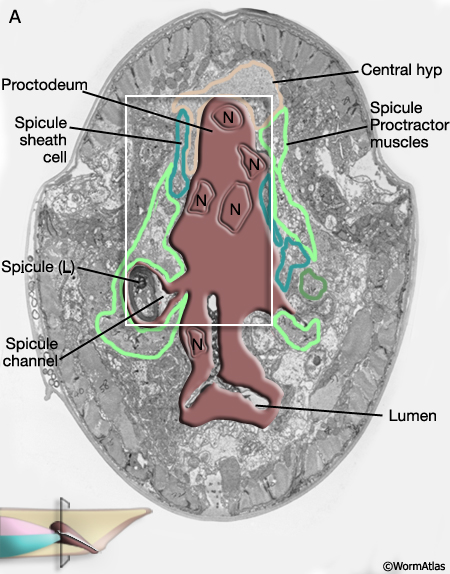
MaleProcFIG 9: Late morphogenesis of the proctodeum. A-D show transverse views moving posteriorly through the proctodeum of a late stage L4 male. Below each diagram is a corresponding TEM section. Clicking on the micrograph image will open a new window with a higher resolution, annotated version of the micrograph. Bε = B.al/rap. (dsr) Dorsal retractor muscles; (Rec gl.) rectal gland cells; (PCS) post-cloacal sensilla. (Adapted from Sulston et al., 1980 and based on the JSG EM series.)
|
MaleProcFIG 10: Epifluorescent views of cells in the developing proctodeum. A. Lateral, left medial plane of late L4 stage. Epifluorescent images from transgenic animals expressing the cog-1::GFP reporter gene. (Strain source: R.E. Palmer and P.W. Sternberg.) B. Lateral, left sublateral plane of late L4 stage. Epifluorescent images from transgenic animals expressing the egl-17::GFP reporter gene. (Strain source: R.D. Burdine and M.J. Stern.) (Cell identification by L.I. Jiang and P.W. Sternberg.)
3.2 Proctodeum of the Adult
The L4 and adult proctodeum differ in a number of ways. The overall shape of the cloaca changes, possibly due to the attachment of muscles and to the general reduction of tail volume associated with tail remodeling. The various cuticles of the proctodeum (lumenal, spicule and gubernacular), initially secreted during L4, become substantially thickened by the adult stage. The cloacal and spicule channel cuticle is presumably secreted by cells lining the lumen. Spicule cuticle is secreted by the spicule socket cells (Jiang and Sternberg, 1999). The gubernaculum is produced by B.paa, with contributions from Bδ(l/r) (Sulston et al., 1980). Cell boundaries in the adult become confused or disappear, making it difficult to follow the profile of individual epithelial cells (MaleProcFIG 11, 12). Many cells contain numerous vesicles, mitochondria, ER and ribosomes consistent with their likely role in the production of cuticle. Some cells, particularly those of the dorsal anterior cloaca, contain large vacuoles. There may also be some repositioning of nuclei in the adult. For example, nuclei lateral to the spicule channels in L4 are absent from this region in the adult. These nuclei may be displaced medially by the attachment of the spicule protractor muscles, which causes substantial compression of the epithelia in this region (see below). Several proctodeal cells, particularly those dorsal and medial to the spicule channels, contain 2-4 nuclei or possibly more (MaleProcFIG 11B). At least one example of cell fusion was previously noted (F.rd and F.ld) by Sulston et al. (1980). The largest syncytium appears to be continuous with the epithelia that line the spicule channels. In MaleProcFIG 11B, dark-staining cell processes lie beneath the spicule protractor muscles and line the spicule channels. These processes are continuous with dark-staining cells in the dorsal proctodeum that contain multiple nuclei.
 Several classes of sex-specific muscles are associated with proctodeum: the spicule retractors and protractors, the anal depressor, the gubernacular erector and retractor muscles (MaleProcFIG 12A-D; MaleProcFIG 9A-D). These muscles control spicule movement or the positioning of the gubernaculum. The muscles are linked to cloacal or spicule channel cuticle or to the spicules or gubernaculum. These muscle-cuticle connections involve hemidesmosomes (Hd) that span the epithelia and are linked to dense bodies in the muscle (see in Male - Muscle System).
MaleProcFIG 12A&B: Proctodeum-associated muscle groups. A. Diagrams illustrating the location of the muscles associated with the proctodeum. B. Low-power TEM showing the location of the spicule protractor muscles and the gubernaculum within the proctodeum, transverse view. Regions marked by white squares depicted in MaleProcFIG 12C&D, respectively. (Image source: N2Y [MRC] 45.)
MaleProcFIG 12C&D: Proctodeum-associated muscle groups. C. TEM, transverse view of spicule channel with surrounding muscles. (Image source: N2Z [MRC] 913-17.) D. TEM, transverse view highlighting the cloacal lumen and surrounding tissues. (Image source: N2Z [MRC] 913-17.)
As described above the connection between the cloaca and gonad is formed by U descendents, K.a, K' and Bε(l/r).aav (MaleProcFIG 13A, B, C). Bε(l/r).aav and its anterior neighbor, the most posterior cell of the vas deferens, are synaptic targets of post-cloacal sensilla (PCBL/R and PCCL/R) and spicule (SPCL/R) neurons. Bε(l/r).aav (and the vas deferens cell) are characterized by the presence of numerous darkly-staining vesicles suggesting they may be secretory cells (MaleProcFIG 13D). The PCS and spicule function in the steps that precede ejaculation, vulval location and spicule insertion. Direct gonadal innervation by SPCL/R, PCBL/R and PCCL/R suggests that vulval contact and fully extended spicules may trigger this step (reviewed in WormBook: Male Mating Behavior).
MaleProcFIG 13A: Cloacal-gonad connection. Diagram, lateral view. U.lp or U.rp may fuse with U.ra or U.la, respectively to form a binucleate cell, see MaleProcFIG 4. (Rec.gl) Rectal gland; (Sph) sphincter muscle. (Based on the N2Y [MRC] LP EM series.)
MaleProcFIG 13B&C: Proctodeum connecting cells. B. Low-power TEM showing the connection between the cloaca and gonad as formed by the descendents of U, transverse view. * or product of their fusion with U. (l/r)a. (Image source: N2Y [MRC] 83.) C. Higher magnification TEM of white boxed region in B, transverse view. (Image source: N2Y [MRC] 83.)
MaleProcFIG 13D: Dark-staining vesicles suggest secretory function for Bε(l/r).aav and vas deferens cell. TEM, transverse view. (Image source: N2Y [MRC] PAG 980.)
At the junction of the vas and cloaca the epithelium appears to have a closed lumen that is surrounded by septate junctions (MaleProcFIG 13D). We speculate that this lumen can reversibly open by the unzipping of junctions and unfolding cell borders, possibly in response to synaptic inputs from the dorsal PAG. A similar set of reversible septate junctions has been suggested to control the opening and closing of the spermatheca in the hermaphrodite reproductive system (see SomaticFIG 9; Hermaphrodite Reproductive System - Somatic Gonad).
4 List of Cells of the Proctodeum (See BYUF lineage)
B lineage cells of the adult cloaca and spicule channels:
Bα.lv (where Bα=B.alaa or B.araa)
Bα.rv
Bγ.alv (where Bγ=B.alpp or B.arpp)
Bγ.arv
Bγ.ald or Bγ.ard (1 of these cells dies)
Bδ.l (where Bδ=B.alpp or B.arpp)
Bδ.r
Bεl.aad (where Bε=B.alap or B.arap)
Bεr.aad
Bεl.aav or Bεr.aav (1 of these cells dies)
Bεl.apa
Bεr.apa
Bεl.app
Bεr.app
Bεl.pv
Bεr.pv
B.paa
B.pap
B.pppa
F lineage cells of the adult cloaca and spicule channels:
F.ld
F.rd
F.lvv
F.rvv
K lineage cells of the adult cloaca:
K.a (ABplpapppaaa)
K' (ABplpapppap)
P12 lineage cells of the adult cloaca:
P12.pa (hyp12) (ABp(l/r)apappapa)
U lineages cells of the adult cloaca:
U.lp
U.la (may fail to divide and fuse with U.lp)
U.rp
U.ra (may fail to divide and fuse with U.lp)
Y lineage cells of the adult cloaca:
PChL (Y.plaa)
PChR (Y.praa)
Spicule cells of the adult:
1. Spicule sheath (SPsh) cells (cells of the right and left fused to form 2-cell syncytiums)
SPshDL (B.alpapap/Bζ(l).pap)
SPshVL (B.a(l/r)aaldp/Bβ.ldp)
SPshDR (B.arpapap/Bζ(r).pap)
SPshVR (B.a(l/r)aardp/Bβ.rdp)
2. Spicule socket (SPso) cells (cells of the right and left fused to form 4-cell syncytiums)
SPso1L (B.a(l/r)pppl/Bγ.pl)
SPso2L (B.a(l/r)aald/ Bα.ld)
SPso3L (B.a(l/r)aalv/Bβ.lv)
SPso4L (B.alpapp/Bζ(l).pp)
SPso1R (B.a(l/r)pppr/Bγ.pr)
SPso2R (B.a(l/r)aard/Bα.rd)
SPso3R (B.a(l/r)aarv/Bβ.rv)
SPso4R (B.arpapp/Bζ(r).pp)
3. Spicule neurons
SPDL (B.alpapaa/Bζ(l).paa)
SPDR (B.arpapaa/Bζ(r).paa)
SPVL (B.a(l/r)aalda/Bβ.lda)
SPVR (B.a(l/r)aarda/Bβ.rda)
SPCL (B.alpaap/Bζ(l).ap)
SPCR (B.arpaap/Bζ(r).ap)
Rectal Epithelial Blast Cells (L1 male)
B
F
Y
U
K'
K
Intestinal-Rectal Valve Cells (Larval and adult male)
virL
virR
Rectal Gland Cells (Larval and adult male)
rectD
rectVL
rectVR
5 References
Baird, S.E. and Ellazar, S.A. 1999. TGFbeta-like signaling and spicule development in Caenorhabditis elegans. Dev. Biol. 212: 93-100. Article
Chamberlin, H.M. and Sternberg, P.W. 1993. Multiple cell interactions are required for fate specification during male spicule development in Caenorhabditis elegans. Development 188: 297-324. Article
Chamberlin, H.M. and Sternberg, P.W. 1994. The lin-3/let-23 pathway mediates inductive signalling during male spicule development in Caenorhabditis elegans. Development 120: 2713-21. Article
Garcia, L.R., Mehta, P. and Sternberg, P.W. 2001. Regulation of distinct muscle behaviors controls the C. elegans male's copulatory spicules during mating. Cell 107: 777-788. Article
Herman, M.A. and Horvitz, H.R. 1994. The Caenorhabditis elegans gene lin-44 controls the polarity of asymmetric cell divisions. Development 120: 1035-47. Article
Jiang, L.I. and Sternberg, P.W. 1999. Socket cells mediate spicule morphogenesis in Caenorhabditis elegans males. Dev. Biol. 211: 88-99. Article
Liu, K.S. and Sternberg, P.W. 1995. Sensory regulation of male mating behavior in Caenorhabditis elegans. Neuron 14: 79-89. Article
Sternberg, P.W. and Horvitz, H.R. 1988. lin-17 mutations of Caenorhabditis elegans disrupt certain asymmetric cell divisions. Dev. Biol. 130: 67-73. Abstract
Sulston, J. 1988. Cell lineage. In The nematode C. elegans (ed. W.B. Wood). Chapter 5. pp 123-155. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York.
Sulston, J.E. and Horvitz, H. R. 1977. Post-embryonic cell lineages of the nematode Caenorhabditis elegans. Dev. Biol. 56: 110-156. Article
Sulston, J.E., Albertson, D.G. and Thomson, J.N. 1980. The Caenorhabditis elegans male: Postembryonic development of nongonadal structures. Dev Biol. 78: 542-576. Article
Sulston, J.E., Schierenberg, E., White J.G. and Thomson, J.N. 1983. The embryonic cell lineage of the nematode Caenorhabditis elegans. Dev. Biol. 100: 64-119. Article
|

This chapter should be cited as: Lints, R. and Hall, D.H. 2009. Male alimentary system, proctodeum. In WormAtlas. doi:10.3908/wormatlas.2.2
Edited for the web by Laura A. Herndon. Last revision: July 24, 2013. |
|