
|
|
|
THE AGING EXCRETORY SYSTEM
 Click pictures for new window with figure and legend, click again for high resolution image Click pictures for new window with figure and legend, click again for high resolution image
1 General Description
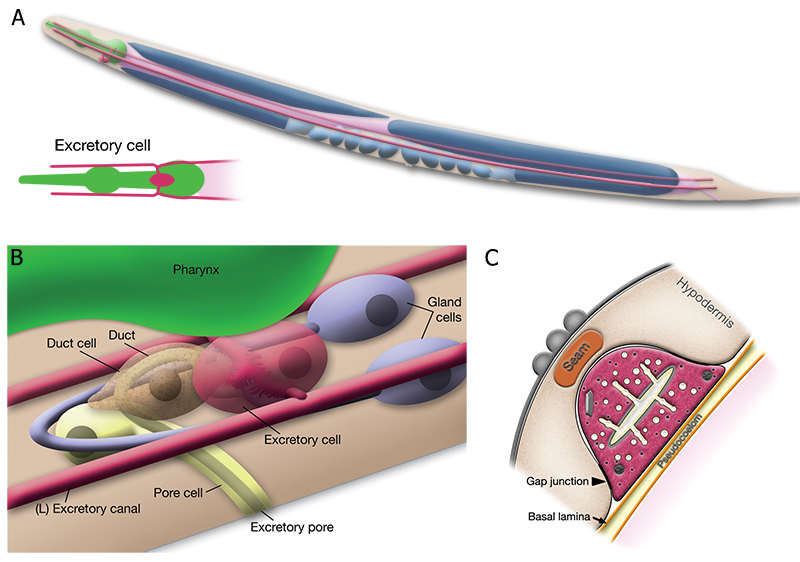
The C. elegans excretory system performs the functions of osmotic regulation, fluid balance, waste filtration and excretion. (See also Hermaphrodite Excretory System). Four cell types make up the excretory system (AExcFIG 1) (Nelson et al., 1983). A single large excretory cell (also called the canal cell) extends long lateral processes along the bodys length to absorb and transport waste to the main excretory duct and pore located in the head, on the ventral side of the pharynx terminal bulb. The excretory cell filters fluid for excretion to the outside of the animal, serving a function similar to the mammalian kidney and ureter structures. The pore cell, the duct cell and bilateral gland cells constitute the other parts of the excretory apparatus. The bilateral gland cells fuse together at the excretory sinus, near the excretory pore. The gland cell functions are currently not well known. The gland cell cytoplasm contains large secretory granules whose contents can be released into the secretory duct and pore during molting (Hall and Hedgecock, 1991).
This simple system must remain intact for the animals viability, as defects which interrupt the patency of the excretory outflow will lead to a rapid swelling of the animal with excess fluid, known as the lethal rod phenotype (Sundaram and Buechner, 2016). Although that mutant phenotype most often arises in early larval development, by disconnection of the pore and duct cells to one another, we presume this passageway must remain at least partially functional for the full life of the animal.
2 Aging of the Excretory Cell
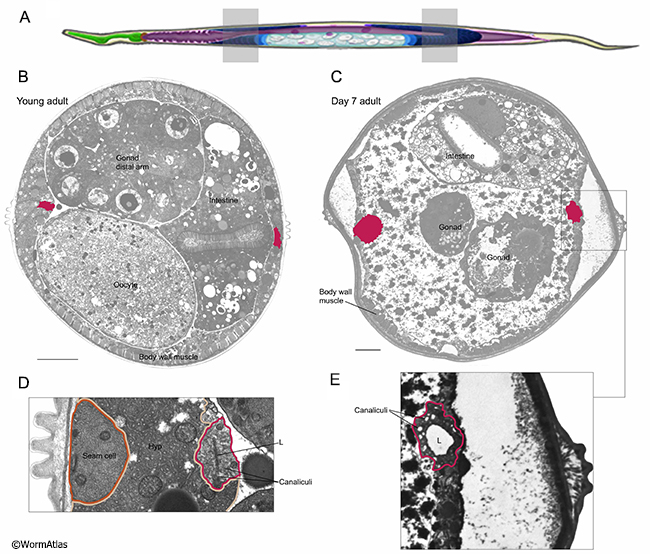
In young animals, the excretory cell forms long bilateral tubes that extend lengthwise along the body axis from nose to tail (AExcFIG 1). These tubes contain a central lumen, or canal, surrounded by multiple smaller canaliculi (see also ExcFIG 7B&C). The canaliculi most likely concentrate and transport materials from the hypodermis and extracellular pseudocoelum to the central canal for excretion. The longitudinal canals are tightly coupled to the lateral hypodermis by a series of very large gap junctions along their length, which are presumed to allow flow of solutes and waste products from the hypodermal cytoplasm into the excretory canals (AExcFIG 3A) (see also ExcFIG 7B&C) (Hall, 2016). The excretory canal pulsates in a rhythmic fashion to transport its contents to the pore and duct for excretion. Excretory canal pulses have been observed in young adults and developmentally-arrested dauer larvae (Nelson et al., 1983). Excretory canal motions in old adults have not been examined.
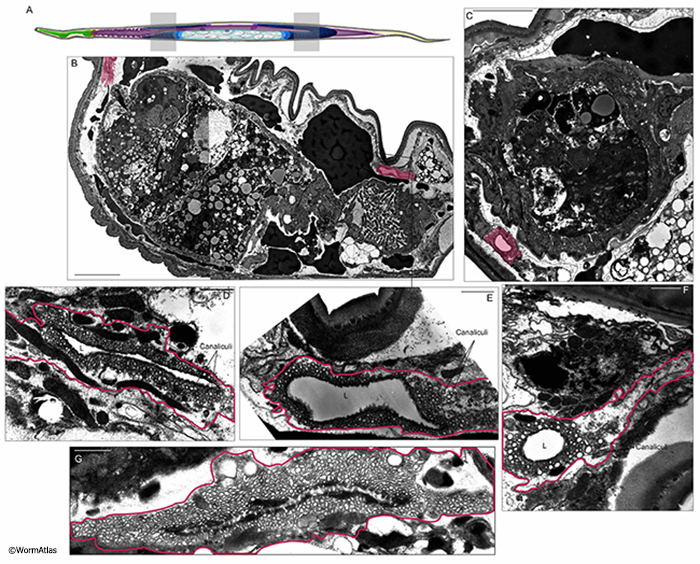
Structurally the excretory cell in aged adults seems to remain intact, though it becomes more disorganized in appearance, most often with a moderately swollen lumen (Herndon et al., 2002; 2017). Defects are generally much worse as the animals get older (AExcFIG 2, AExcFIG 4, AExcFIG 5 & AExcFIG 6). In some animals, the apical membrane facing the lumen sometimes becomes excessively thickened and more electron dense (AExcFIG 2G&I). The cell cytoplasm can become less electron dense, with dark inclusions and sometimes containing debris, damaged organelles or massive swollen vacuoles. In some cases, the number of canaliculi appears to decline with age and the excretory cell may contain multiple lumens or perhaps branched lumens (AExcFIG 2 & AExcFIG 4). As has been previously seen in younger adults (Buechner et al., 1999; Altun and Hall, 2009, Kolotuev et al., 2013), the shape of canaliculi can vary between a narrower smooth tube, to a string of connected “beads”, to larger diameter disconnected beads. In these aged adults, this latter phenotype seems to predominate, especially at 15 days, and may indicate lessened opportunity for efficient excretion. At the basal border, facing the pseudocoelom or where becoming detached from the hypodermis, the excretory canals can appear ruffled in older adults. Integrity of the gap junctions between the canals and the hypodermis is not obvious in aging animals, and the hypodermis is often more impacted by degeneration than the canals. Indeed the canal sometimes seemingly detaches from the aging hypodermis as the latter tissue degrades and shrinks (AExcFIG 3). These changes are heterogeneous and sometimes the excretory cell may appear normal in other older adults, or one canal can appear more functional than its bilateral partner (Herndon et al., 2017). It is not currently known if aging affects the excretory cell differently along its length, or if similar structural changes extend along the excretory cell from head to tail.
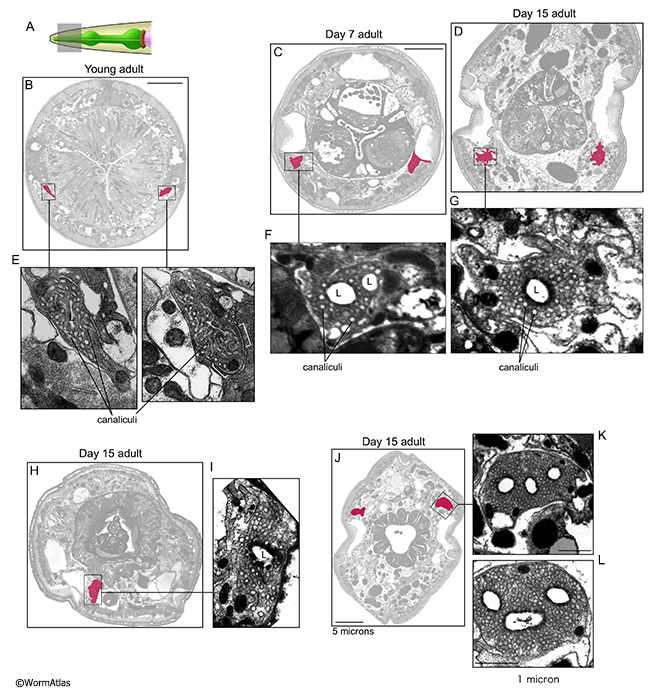
AExcFIG 4: Excretory cell structure near the posterior pharynx during aging. A. Diagram of the C. elegans head showing the approximate positions of photomicrographs B,C&D by shaded box in the posterior portion of the pharynx adjacent to the pharygeal-intestinal valve. B,C,D. Transverse sections from a young adult (B) and two day 15 adults (C&D). The bilateral excretory canals are colored in pink. The apical borders are smooth in the young adult, becoming disorganized and ruffled in both day-15 adults. Lower panels, closeups of individual excretory canals as shown. One excretory cell (D) contains two lumens, the other cells contain one lumen. D. Both excretory canals appear to contain fewer canaliculi than the other animals. C. Bar, 5 microns. (Image source: B: N513A [D. Hall] G534 M1 959, C: N829 [D. Hall] W001, D: N829 [D. Hall] R128).
Another significant change in the functioning of the excretory cell is not reflected in the changes in canal structure, but in the progressive distortion of the pseudocoelomic cavity in aging animals. Much of this enlargement is filled with yolk protein, but there is also a substantial increase in fluid here, at least 10-fold as estimated from the LM movies, and supported by the huge swelling noted in transverse thin sections of the aging animals (AExcFIG 5C; AExcFIG 7C).
A variety of mutations are known to result in swollen or cystic phenotypes in developing excretory canals that mimic some of the features that are noted in the aging process in wild type canals (AExcFIG 2). In particular many of the exc mutant alleles clearly cause dramatic changes at the apical membrane surrounding the canal lumen (Buechner et al., 1999). This might suggest that the aging process includes a decline in the functions of these gene products over time. For instance, loss of expression of several intermediate filament proteins, EXC-2 and IFA-4, leads to cysts, while their overexpression is known to cause a narrowing of the lumen (Al-Hashimi et al., 2018). These proteins are thought to form part of the terminal web along the cytoplasmic surface of that apical membrane, which is presumed to reinforce the membrane in the normal excretory canal. The TEM images of the aging canal show an increase of electron density lining the cytoplasmic side, near to where one normally expects to find the terminal web as a separate feature (AExcFIG 2G,J). This suggests the intermediate filament components of the terminal web may have collapsed onto the membrane surface as part of the aging process. Several more mutants are involved in endosomal trafficking events at the apical membrane, including EXC-5 and EXC-1 (Mattingly and Buechner, 2011; Grussendorf et al., 2016). The status of the canals apical membrane is also sensitive to interplay between two membrane proteins, ERM-1 and AQP-8, controlling water flux across the apical membrane. Cystic phenotypes can result when these proteins are not present in proper proportions during early development (Khan et al., 2013), and perhaps also during aging.
Another set of genes, LET-653, LPR-1, LPR-3, help to organize the extracellular matrix lining the luminal side of the apical membrane (cf. Gill et al., 2016; Forman-Rubinsky et al., 2017). Cell autonomous loss of exc gene function within this single cell during aging might weaken maintenance of the apical membrane, producing disease-like conditions.
3 Aging of the Excretory Gland Cell
The excretory gland cell is a binucleate cell located in the head near the pharynx terminal bulb (see ExcFIG 3C; Hermaphrodite Excretory System). The gland cell’s nuclei are contained in separate cell bodies positioned on the left and right sides of the body. These cell bodies are connected by a pair of anteriorly projecting processes that extend toward the nerve ring, connecting twice to form an A-shaped structure (Nelson et al., 1983). Some regions of the gland cell bodies and processes contain dark secretory granules, which have been noted to move from cell body into the cell processes during molting (Hall and Hedgecock, 1991).
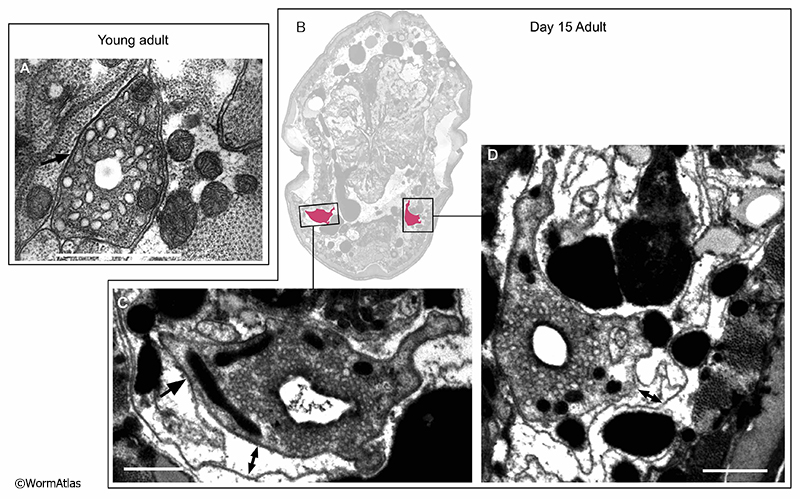
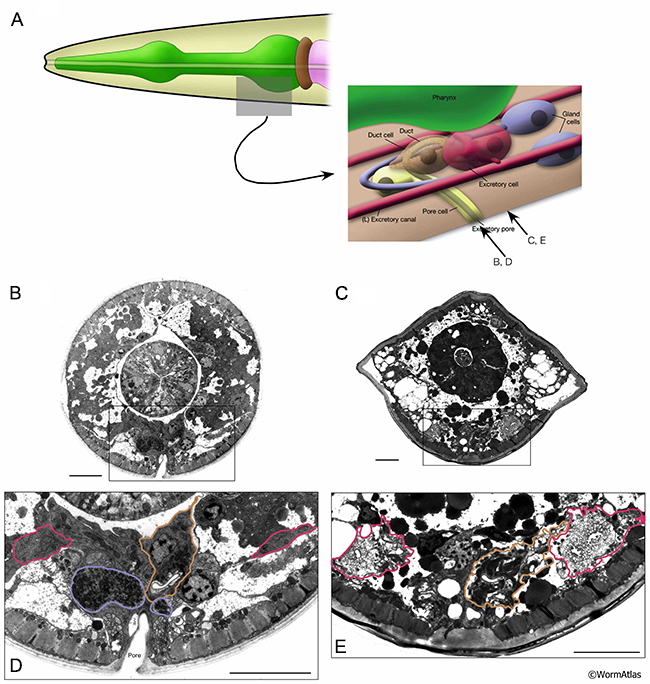
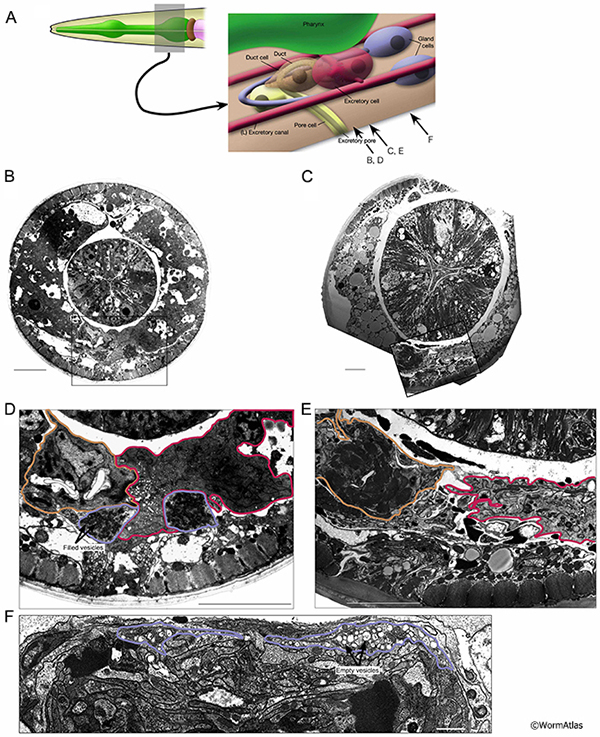
In aged adults, gland cell processes often become disorganized and more difficult to identify in the regions adjacent to the excretory cell (AExcFIG 7 & AExcFig 8). Cells containing dark secretory granules may be found in regions where the gland cells would be expected, but are often poorly preserved structurally. The contents of these vesicles can be altered, becoming less electron dense or sometimes seemingly empty (AExcFig 8F). Overall, it appears that secretory granules are less predominant in the gland cells of older adults. Alternatively, the gland cell processes may deteriorate during aging, making them less distinguishable in microscopy sections. The specialized secretory junction connecting the gland cell to the excretory cell and the excretory duct (see also ExcFIG 8) has thus far not been found in any of the aging animals, though this junction might still be found by further studies. However, survival of the aging adult may not depend on gland cell functionality, as the gland cells are thought to be needed for molting during larval development.
4 Aging of the Excretory Pore and Duct
In young adults, the duct and pore cells compose and enclose a cuticle lined pore that opens to the exterior environment (see ExcFIG 9; Hermaphrodite Excretory System). A specialized, porous “secretory junction” connects the excretory canal cell lumen, the excretory gland cells, and the excretory duct (see ExcFIG 8). The duct and pore are sometimes still recognizable in micrographs of older adults, although the detailed pathway of these cells have not been traced in old age (AExcFIG 7 & AExcFig 8). The decline of surrounding tissues during aging make the smaller duct and pore quite difficult to discern in many cases compared to the canal cell or its soma. As mentioned above, many adult animals develop a swollen appearance as they age, suggesting some defects affect excretory system functions during aging. Whether this occurs at the level of transport within the excretory canal or at the level of export from the body at the duct and pore remains to be investigated.
|
5 References
Al-Hashimi, H., Hall, D.H., Ackley, B.D., Lundquist, E.A. and Buechner, M. 2018. Tubular excretory canal structure depends on intermediate filaments EXC-2 and IFA-4 in Caenorhabditis elegans. Genetics 10.1534/genetics.118.301078. Article
Altun, Z.F. and Hall, D.H. 2009. Excretory system. In WormAtlas. doi:10.3908/wormatlas.1.17
Buechner, M., Hall, D.H., Bhatt, H. and Hedgecock, E.M. 1999. Cystic canal mutants in Caenorhabditis elegans are defective in the apical membrane domain of the renal (excretory cell). Dev. Biol. 214: 227-241. Article
Forman-Rubinsky, R., Cohen, J.D. and Sundaram, M.V. 2017. Lipocalins are required for apical extracellular matrix organization and remodeling in Caenorhabditis elegans. Genetics 207: 625-42. Article
Gill, H.K., Cohen, J.D., Ayala-Figueroa, J., Forman-Rubinsky, R., Poggioli, C., Bickard, K., Parry, J.M., Pu, P., Hall, D.H. and Sundaram, M.V. 2016. Integrity of narrow epithelial tubes in the C. elegans excretory system requires a transient luminal matrix. PLOS Genetics 12: e1006205. Article
Grussendorf, K.A., Trezza, C.J., Salem, A.T., Al-Hashimi, H., Mattingly, B.C., Kampmeyer, D.E., Khan, L., Hall, D.H., Göbel, V., Ackley, B.D. and Buechner, M. 2016. Facilitation of endosomal recycling by an IRG protein homologue maintains apical tubule structure in Caenorhabditis elegans. Genetics 203: 1789-1806. Article
Hall, D.H. 2016. Gap junctions in C. elegans: their roles in behavior and developmentl. Dev. Neurobiol. doi: 10.1002/dneu.22408. Article
Hall, D.H. and Hedgecock, E.M. 1991. Kinesin-related gene unc-104 is required for axonal transport of synaptic vesicles in C. elegans. Cell 65: 837-847. Abstract
Herndon, L.A., Schmeissner, P.J., Dudaronek, J.M., Brown, P.A., Listner, K.M., Sakano, Y., Paupard, M.C., Hall, D.H. and Driscoll, M. 2002. Stochastic and genetic factors influence tissue-specific decline in ageing C. elegans. Nature 419: 808-814. Article
Herndon, L.A., Wolkow, C.A., Driscoll, M. and Hall, D.H. 2017. Effects of ageing on the basic biology and anatomy of C. elegans. In Ageing: lessons from C. elegans. (ed Olsen, A. and Gill, M.). Chapter 2. pp. 9-39. Springer International, Switzerland. Abstract
Khan, L.A., Zhang, H., Abraham, N., Sun, L., Fleming, J.T., Buechner, M., Hall, D.H. and Gobel, V. 2013. Intracellular lumen extension requires ERM-1-dependent apical membrane expansion and AQP-8-mediated flux. Nat. Cell Biol. 15: 143-56. Article
Kolotuev, I., Hyenne, V., Schwab, Y., Rodriguez, D. and Labouesse, M. 2013. A pathway for unicellular tube extension depending on the lymphatic vessel determinant Prox1 and on osmoregulation. Nature Cell Biol. 15: 157-68. Abstract
Mattingly, B.C. and Buechner, M. 2011. The FGD homologue EXC-5 regulates apical trafficking in C. elegans tubules. Dev. Biol. 359: 59-72. Article
Nelson, F.K., Albert, P.S. and Riddle, D.L. 1983. Fine structure of the Caenorhabditis elegans secretory-excretory system. J. Ultrastruct. Res. 82: 156-171. Article
Sundaram, M.V. and Buechner, M. 2016. The Caenorhabditis elegans excretory system: A model for tubulogenesis, cell fate specification, and plasticity. Genetics. 203:35-63. Article
* Description of Behavioral Classes (A, B, C) as described in Herndon et al., 2002
To characterize aging phenotypes, age-synchronized individual worms were scored both for spontaneous movement and for response to prodding with a wire over the course of their lifespan. Three distinct classes representing behavioral phenotypes were established. Animals that move constantly and make sinusoidal tracks were designated as class A. Class B animals mainly move when prodded. When they move it is with uncoordinated motion, leaving non-sinusoidal tracks. Class C animals do not move forward or backward, even upon prodding, but do show head and/or tail movement and twitch in response to touch. All animals begin adulthood in class A. Class B animals appear around days 6-7 of adulthood and class C around day 9-10 (at 20oC). At later ages, animals representing all classes can be found within the same population and it was found that the behavioral class type was the better predictor of life expectancy than chronological age (Herndon et al., 2002). Due to the stochastic nature of aging in an individual nematode, these classifications only reflect ongoing changes in nerve and muscle, while other tissues can show very different age-related effects within one behavioral class, declining faster or remaining healthy much longer.
|

This chapter should be cited as: Wolkow, C.A., Herndon, L.A. and Hall, D.H. 2019. The Aging Excretory System. In WormAtlas. doi:10.3908/wormatlas.8.5
Edited for the web by Laura A. Herndon. Last revision: January 4, 2019. |

|
|
|
|